New findings of the mechanisms in the interaction between extracellular vesicles and recipient cells by the groups of Nankai University
A research article entitled “High TSPAN8 expression in epithelial cancer cell-derived small extracellular vesicles promote confined diffusion and pronounced uptake” was published in J Extracell Vesicles by the groups of Associate Professor Shijing Yue from the School of Medicine and Professor Lehui Xiao from the College of Chemistry. The researchers used genetically engineered breast cancer cells to derive TSPAN8-enriched small extracellular vesicles (sEVs) and evaluated the impact of TSPAN8 on target cell membrane’s diffusion and transport properties. The single-particle tracking technique showed that TSPAN8 significantly promoted sEV binding via confined diffusion. Functional assays indicated that the transgenic TSPAN8-sEV cargo increased cancer cell motility and epithelial-mesenchymal transition (EMT). This study is contribute to clarify that TSPAN8 modified sEVs can improve the efficiency of drug delivery system applications.

In recent years, sEVs have been widely used in the diagnosis and therapy of diseases due to their specific advantages. A large number of biologically active molecules in sEVs package that can be transported to various organs and tissues of the body through the bloodstream. SEVs play a key regulatory role in cell-to-cell communication, and sEVs cargos may affect the phenotype and function of recipient cells. Studies have shown that sEVs cargos can regulate the phenotype of target cells in the physiological or pathological microenvironment. Epithelial cancer cell-derived sEVs enriched in TSPAN8 or CD151 and integrins to form complexes have been shown to promote cancer metastasis by impairing cell adhesion and migration. However, the binding process of sEVs and recipient cells and related molecular mechanisms are still unclear.
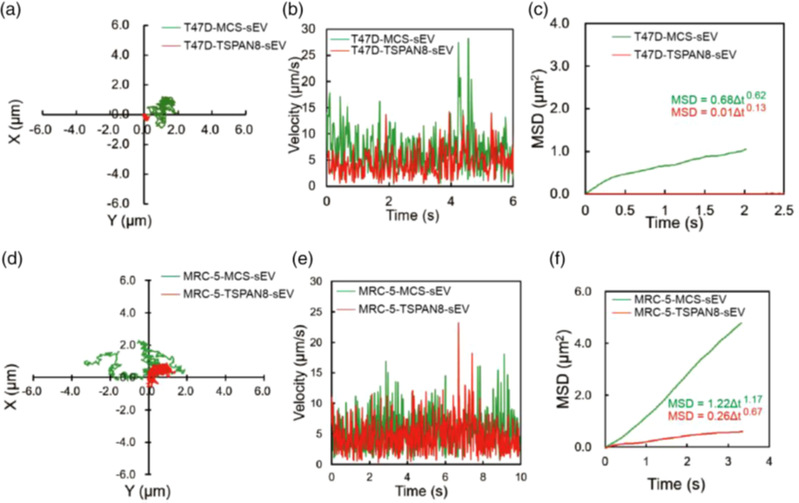
TSPAN8 changed sEV diffusion mode to target cells
This study proved for the first time that TSPAN8 molecules can promote the confined diffusion of sEVs and pronounced uptake by recipient cells, thereby promoting tumor cell migration and EMT and inducing tumor metastasis. In vivo, transgenic TSPAN8-sEV promoted uptake of sEVs in the liver, lung, and spleen. We concluded that TSPAN8encourages the sEV-target cell interaction via forced confined diffusion and significantly increases cell motility. Therefore, TSPAN8-sEV may serve as an important direct or indirect therapeutic target.
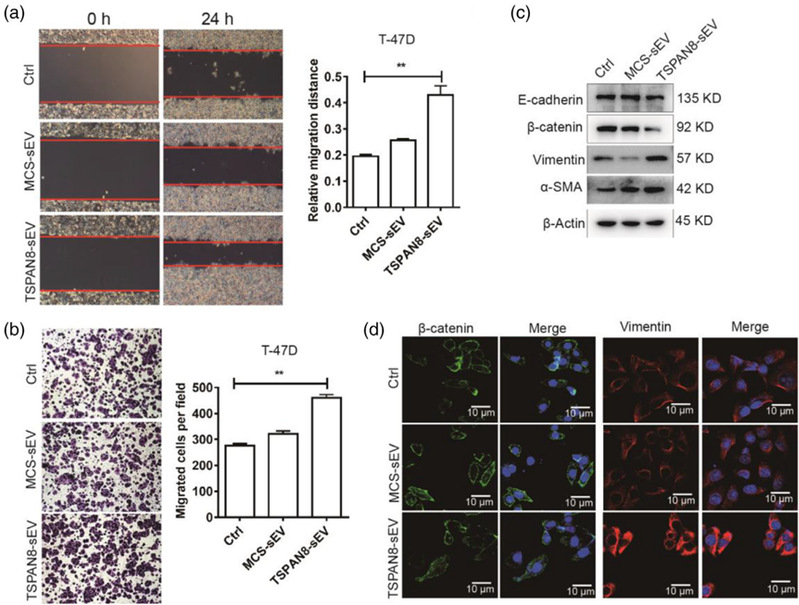
sEV cargo TSPAN8 educating recipient cells activation
The first authors of this article are Teng Wang, a master student from the School of Medicine, Xin Wang, a doctoral student from the College of Chemistry, and Haobin Wang, a deputy director doctor from the Third People’s Hospital of Chengdu. The master students of Luhan Li and Chenhong Zhang are deeply involved in this research. Professor Lehui Xiao from the College of Chemistry and Associate Professor Shijing Yue from the School of Medicine are the co-corresponding authors, and Nankai University is the first Institute of completion.
The research was funded by the National Natural Science Foundation of China.
The full text link: https://onlinelibrary.wiley.com/doi/10.1002/jev2.12167





